
According to the public relations report of Sharif University of Technology, Dr. Alireza Mushfaq, a member of the Faculty of Physics of Sharif University of Technology, with the cooperation of a group of researchers from Sharif University, South Korea's SKKU and Al-Zahra University, succeeded in solving a challenging problem to reduce the consumption of carbon materials in the field of energy production. Publish yourself in the prestigious journal ACS Catalysis.
This article titled "Hierarchical nickel oxide microspheres based on an array of nanorods as a dual-function electrocatalyst for photolysis/selective electrolysis and resistant to seawater corrosion", by theoretically and experimentally solving a challenging problem to reduce the consumption of carbon materials in The field of energy production now promises the use of abundant and clean resources on the planet (photovoltaic technology in seawater).
It is stated in this article; One of the main challenges facing humanity in the 21st century is the energy supply crisis. About 85% of the world's total energy consumption is provided through the use of fossil fuels, which, in addition to the fact that their resources are limited, will not meet human energy needs in the coming years; They cause the emission of a large amount of greenhouse gases, including carbon dioxide. Following the emission of greenhouse gases in the space, climate changes are taking place, including changes in the amount of annual rainfall and changes in the pH of sea water, which threaten human health. In fact, the harmful environmental effects are expanding in a large way, and with the continuation of the current conditions of the planet Earth, it will move towards unacceptable conditions for future generations. To solve these problems, fundamental developments in the field of energy and society's acceptance to consume less carbon materials are very necessary. Therefore, the importance of using clean and renewable sources as a suitable and potential alternative to fossil fuels is more mandatory.
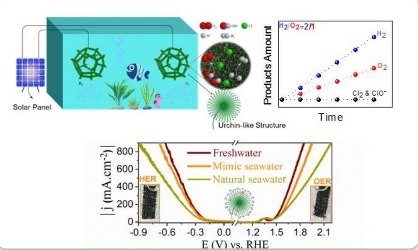
Hydrogen is a clean and storable, sustainable and environmentally friendly energy carrier that can be produced through water electrolysis and is effective in combating climate change and achieving zero greenhouse gas emissions. Because the cycle of production, consumption and regeneration of hydrogen is done without carbon emission. But the production of hydrogen through the decomposition of pure water will put a lot of pressure on the world's fresh water resources. Therefore, the dependence of water electrolysis technology on fresh water resources will be a great threat to the sustainable environment. On the other hand, the salty and low-quality waters in the oceans and seas are one of the many resources on the planet that can be economically used to resolve climate change and provide clean energy, so that the electrolysis of sea water for production Sustainable hydrogen and bioremediation can become an attractive and flexible technology. In fact, seawater electrolysis technology is reminiscent of the saying "kills two birds with one stone" because it can be used both for hydrogen production and for desalination of seawater. Electrocatalysts are considered as the key component of electrochemical systems of seawater decomposition. Therefore, the development and use of suitable electrocatalysts, whose materials are economically viable and have good stability against seawater corrosion, and ultimately show better efficiency and performance in the field of providing clean energy and a healthy environment, is a very important thing. It is essential.
Now, in a pioneering research, a group of researchers from Sharif University of Technology, with the efforts of Khadija Hemmati (Faculty of Physics) under the supervision of Dr. Alireza Moshfaq (Faculty of Physics) and the collaboration of researchers from SKKU University in South Korea, under the supervision of Dr. Hiuyang Lee (Faculty of Energy) and Dr. Moradlou from Al-Zahra University (Faculty of Chemistry), theoretically and experimentally succeeded in designing and manufacturing stable and biocompatible electrocatalysts with low costs with dual function for the efficient and durable decomposition of seawater into hydrogen and oxygen fuels, as well as weakening and controlling existing competitive and disturbing reactions. They drowned in sea water. In summary, nickel-based nanostructured electrocatalysts include nickel oxide microspheres, so that the surface of these microspheres is completely uniformly and densely covered by an array of rod-shaped nanostructures, which will lead to a 3D hierarchical structure with a dandelion-shaped morphology. . The presence and participation of a high effective surface with many active sites due to the formation of this architecture, improving the intrinsic activity of each active site, as well as the ability to transfer effective charge due to good electrical conductivity, increase the electrocatalytic activity of the developed system in order to perform both half-reactions of hydrogen gas production. and oxygen and weakening and controlling disturbing and competitive reactions in sea water. The good stability of the synthesized electrocatalyst can be attributed mainly to the corrosion resistance of the nickel oxide hierarchical structure. The results of this research not only suggest the possibility of using non-noble metal electrocatalysts to produce hydrogen and oxygen fuels from sea salt water, but also a new approach for the rational and effective design of structures with 3D hierarchical architecture for use in the field of conversion and Provides energy storage.
This research group also succeeded in setting up an integrated solar seawater decomposition system using the developed electrocatalysts. One of the advantages of hydrogen production using electrocatalytic decomposition of brackish water is its simplicity and economy. For this reason, this system has the ability to be used on a global scale. Hence, the broader perspective of this research is to design and build a device for simultaneous electricity generation.
https://pubs.acs.org/doi/10.1021/acscatal.3c00510
